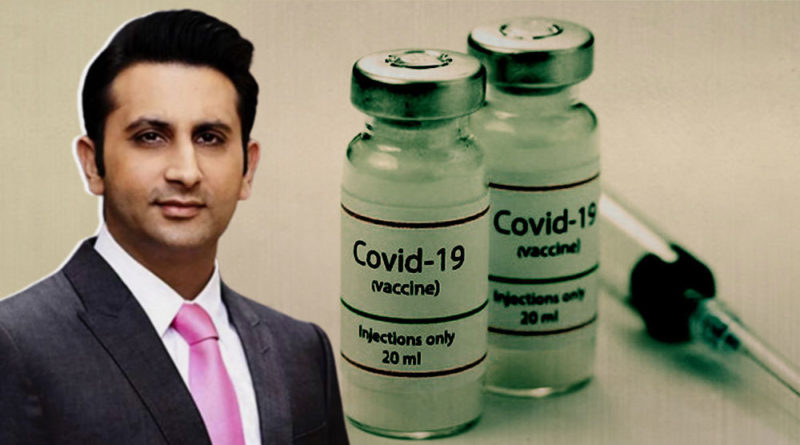
Corona vaccine will be available by April 2021: Serum Institute CEO - Adar Poonawalla
Agencies
New Delhi, 20 Nov 2020: Experts are of the opinion that the coronavirus vaccine can very well be made available by the year 2021. Backing this statement is the Serum Institute of India CEO, Adar Poonawalla. Speaking at the Hindustan Times Leadership Summit, 2020 Poonawalla stated that by February 2021 the Oxford corona-vaccine should be available for the healthcare workers and elderly and by April for the general public. He also maintained that it would be priced at a maximum amount of Rs 1,000 (USD 5-6) per dose.
The Serum Institute asserted that by the year 2024 it was very likely that the entire country would be vaccinated.
"It will probably take two or three years for every Indian to get inoculated, not just because of the supply constraints but because you need the budget, the vaccine, logistics, infrastructure and then, people should be willing to take the vaccine. So these are the factors that lead up to being able to vaccinate 80-90 per cent of the population," he said.
"It will be 2024 for everybody, if willing to take a two-dose vaccine, to be vaccinated." the Serum chief said.
He also said that the required two necessary doses for the public will depend on the final trial results and regulatory approvals.
"The government of India will be getting it at a far cheaper price at around USD 3-4 because it will be procuring the vaccine in a large volume and get access to the price that is similar to what COVAX has got. We are still pricing the vaccine at a far cheaper and more affordable price than other vaccines we have in the market today," Poonawalla said.
Talking about the effectiveness of the said vaccine, Poonawalla stated that the Oxford-Astrazeneca vaccine is working very well in elderly people, which was a concern earlier.
"It has induced a good T-cell response, which is an indicator for your long-term immunity and antibody response but then again, time will only tell if these vaccines are going to protect you in the long term. Nobody can answer that for any of the vaccines today," Poonawalla said.
Meanwhile, on the question regarding the safety of the vaccine, the Serum CEO said that there have been no major complaints, reactions or adverse events. "We would need to wait and see. The efficacy and immunogenicity results from the Indian trials will come out in about a month-and-a-half," he added.
Poonawalla also stated that as soon as the UK authorities and the European Medicines Evaluation Agency (EMEA) approve the emergency use of the vaccine, Serum Institute will apply to the drug controller for emergency use authorisation in India.
"But that will be for a limited use for frontline workers, healthcare workers and elderly people," he added.
Children would have to wait a little longer till the safety data is out, but the good news is that COVID-19 is not so bad and serious for them, Poonawalla said.
"Unlike measles pneumonia, which is deadly, this disease is seeming to be less of a nuisance for children but then, they can be carriers and can give the infection to others.
"We want to vaccinate the elderly people and others who are the most vulnerable first. Once we have enough safety data to go in on children, we can recommend it for children too," he said.
Poonawalla said the Oxford vaccine is affordable, safe and stored at a temperature of two to eight degrees Celsius, which is an ideal temperature for it to be stored in the cold storages of India. He said the SII plans to make about 10 crore doses per month from February.
Referring to the number of doses the country would require, Poonawalla said, "India wants around 400 million doses by July. I do not know if it will take all from the Serum Institute. We are gearing up to offer that kind of volume to India and still have a few 100 million to offer to COVAX by July and August. No agreement so far," he said. Adding to this, he said that the SII is not entering into any agreement for the supply of the vaccine to other countries as India is its priority.
"We have not signed and committed anything else beyond Bangladesh at the moment. We really do not want to partner right now with many countries because we will not have enough stocks to deliver. We want to handle India as a priority first and manage Africa at the same time and then help out other countries," he said.
As per the Serum chief, over 30-40 crore doses of the Oxford vaccine will be available by the first quarter of 2021.
 Write Comment |
Write Comment |  E-Mail To a Friend |
E-Mail To a Friend |
 Facebook |
Facebook |
 Twitter |
Twitter |
 Print
Print